|
Nitrogen bubbles created in human tissue expands when divers ascend to the surface making it dangerous to ascend to quickly or spend too much time underwater. 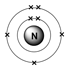
Nitrogen rarely reacts with other chemicals. Properties Nitrogen is a gas at room temperature. Nitrogen is in Period 2 of the Periodic Table because it has 2 electron shells. Whilst inert when breathed in by humans Nitrogen poses several risks to divers through Nitrogen Narcosis and the Bends. Write the atomic number of the element present in the third period and seventeenth group of the. Atomic Structure Nitrogen has 7 protons and 7 neutrons in its nucleus giving it an atomic number of 7 and a atomic mass of 14. It is a non metal gas at room temperature and it has a melting point of -210°c and a boiling point of -195°c. Diagram of the nuclear composition, electron configuration, chemical data, and valence orbitals of an atom of nitrogen-14 (atomic number: 7), the most common. It has an atomic number 7 and an atomic weight of 14.0067 FIGURE 2.3 Lewis dot. Nitrogen was discovered in 1772 by the physician Daniel Rutherford but has been known as a compound in nitric acid and ammonia since the middle ages. Each bond in a triple bond is a pair of electrons, so a triple bond. Nitrogen is widely used in the creation of Ammonia in the Haber process as a fertiliser and is used to make explosives such as TNT. It is especially important in plant development and exists in soil as the compound Ammonia. Despite this, Nitrogen is a vital component in construction of proteins, amino acids and DNA. Nitrogen makes up 78% of the atmosphere and is breathed in and exhaled by humans but is not taken into the human body. Nitrogen is an abundant chemical element that exists in the atmosphere as a diatomic molecule where two atoms of Nitrogen share 3 pairs of electrons in a triple bond. Nitrogen-15 has an atomic number of 7, so an atom of nitrogen-15 contains 7 protons, 7 electrons (recall that the number of protons equals the number of. The total number of electrons in a nitrate. Name: Nitrogen Atomic Number: 7 Element Symbol: N Group: 15 Period: 2 Block: p Element Family: nonmetal Atomic Mass: 14.006 43 14.007 28 IUPAC guidelines. 
It has the atomic number 7 in the periodic table and belongs in Group 15. Nitrogen atom has an atomic number of 7 and oxygen has an atomic number 8. Comprising 79% of the earth atmosphere it is very abundant. Nitrogen (N) exists as a colourless, odourless gas in the earth’s atmosphere.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
